Cement is more and more widely used in our life. Research has proved that the existence of Cl - will cause "electrochemical reaction" in the cement concrete structure, which will lead to steel corrosion and hazard of cement concrete structure. In addition to the "electrochemical reaction", the "oxidation reaction" ,"alkali-orthopaedic reaction" and "acid-alkali corrosion reaction" occurring in the cement concrete structure are also causes that can not be ignored, and Cl- always plays an "inductive" role in the occurrence of these harmful reactions.

The "induction" is mainly caused by the properties of Cl-and the ionic compound (MClx )composed of alkali metal and alkaline earth metal ion (MX+)combined with Cl-. According to the harmful reaction mechanism of chloride ion "induced" cement concrete, the main factors causing this reaction are as follows:
(1) Cl- in cement mainly comes from itself and admixtures added in it. The higher the concentration of Cl- is, the higher the content of MClx is, and the more intense the hazard reaction is. The longer the time, the more serious the damage.
(2) The greater the air humidity or the humidity of the surrounding environment of concrete components, the easier the hazard reaction will occur and the greater the hazard will be.
(3) The higher the ambient temperature is, the more serious the hazard reaction is and the more serious the hazard is.
(4) The longer the time is, the longer the hazard response lasts, and the degree of hazard will gradually expand.
Shenghan Method
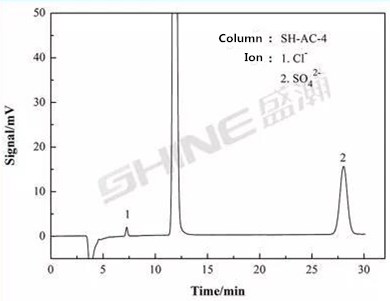
Ion Dhromatograph CIC-D100+Conductivity Detector SW012+Suppressor SHY-A-6

CIC-D100 ion chromatograph, as a classic product of Shenghan, has been widely praised. Based on the latest needs of users, the new CIC-D100 adds classical techniques such as automatic range conductivity detector, which can conveniently test anions, cations and other polar substances in different matrix samples. At the same time, ions with very different concentrations are separated, and the test results are accurate and reliable. The system has fast start-up, reliable and stable performance, and is suitable for a wide range of fields such as pharmacy, environment, food, chemical industry, geology and research laboratories.
After decomposition with nitric acid, the sample solution is made into sample solution, which enters the ion chromatographic column and is eluted by appropriate eluent. The anion is separated because of its different retention characteristics on the column. When it flows through the self-regeneration electrolysis suppressor, the suppressor will reduce the background conductivity of the eluent and increase the conductivity response value of the ions tested. At last, the chromatogram is output after detection by conductivity detector, so that the retention time is qualitative and the peak area (or peak height) is quantitative.
Analysis Conditions
Chromatographic column: SH-AC-4
Eluent: 2.4 mM Na2CO3 + 6.0 mM NaHCO3
Flow velocity: 1.0 mL/min
Column temperature: 35 C
Sampling volume: 25μL
Suppressor current: 75 mA